Get Healthy!
Staying informed is also a great way to stay healthy. Keep up-to-date with all the latest health news here.
12 Sep
Keto Diet Shows Promise in Reducing Depression Among College Students
In a new study, college students with major depressive disorder who followed a keto diet experienced a significant drop in depression symptoms and an increase in overall well-being.
11 Sep
Insomnia May Speed Up Brain Aging and Dementia Risk
A new study suggests chronic insomnia may be an early warning sign or contributor to cognitive decline.
10 Sep
Fall Allergy Survival Guide for Families
Allergies, a cold, or something more? Sorting out the symptoms and treatments with Dr. Farah Kahn, immunologist and allergist at Nationwide Children’s Hospital and a spokesperson for the American College of Allergy, Asthma, and Immunology.
Precise Brain Stimulation May Offer Faster Relief for Depression
A new type of noninvasive brain stimulation may help people with moderate to severe depression feel better faster than standard treatments, researchers in a new report say.
The method, called high-definition transcranial direct current stimulation (HD-tDCS), uses small electrodes on the scalp to deliver mild electrical currents to specific...
- I. Edwards HealthDay Reporter
- |
- September 12, 2025
- |
- Full Page
Protecting Amazon Forests May Also Protect Human Health, Study Finds
Destroying the Amazon rainforest doesn’t just threaten the environment, it may also make people sick.
A study published Sept. 11 in the journal Communications Earth & Environment found that rates of certain diseases were lower in regions where forests were protected and managed by Indigenous peoples.
Research...
- I. Edwards HealthDay Reporter
- |
- September 12, 2025
- |
- Full Page
Nutrient in Vegetables May Help the Body Fight Cancer
A common nutrient found in leafy greens and corn may do more than protect eyesight.
New research shows that zeaxanthin, a plant-derived carotenoid, can strengthen the body’s cancer-fighting immune cells and make immunotherapy more effective.
The study, recently published in the journal Cell Reports Medicine, foun...
- I. Edwards HealthDay Reporter
- |
- September 12, 2025
- |
- Full Page
Can A Keto Diet Help Treat Depression?
A “keto” diet might help people with depression, a small-scale pilot study suggests.
Depression symptoms decreased by about 70% among a small group of college students who followed a ketogenic diet for at least 10 weeks, researchers reported in the journal Translational Psychiatry.
“So many people are suffe...
- Dennis Thompson HealthDay Reporter
- |
- September 12, 2025
- |
- Full Page
Inmates Less Likely To OD After Release If Provided Opioid Addiction Meds In Jail
County jail inmates who receive medication to treat their opioid addiction have a lower risk of overdose after their release, a new study reports.
They’re also significantly more likely to continue their addiction treatment, researchers reported Sept. 10 in The New England Journal of Medicine.
“These findings dem...
- Dennis Thompson HealthDay Reporter
- |
- September 12, 2025
- |
- Full Page
Cervical Cancer Survivors Have Higher Risk of Another Type Of Cancer
Women who overcome cervical cancer might have another potentially life-threatening health crisis on their horizon, researchers say.
Cervical cancer survivors have nearly double the risk of developing anal cancers compared to the general population, researchers reported Sept. 11 in JAMA Network Open.
And their risk increases ...
- Dennis Thompson HealthDay Reporter
- |
- September 12, 2025
- |
- Full Page
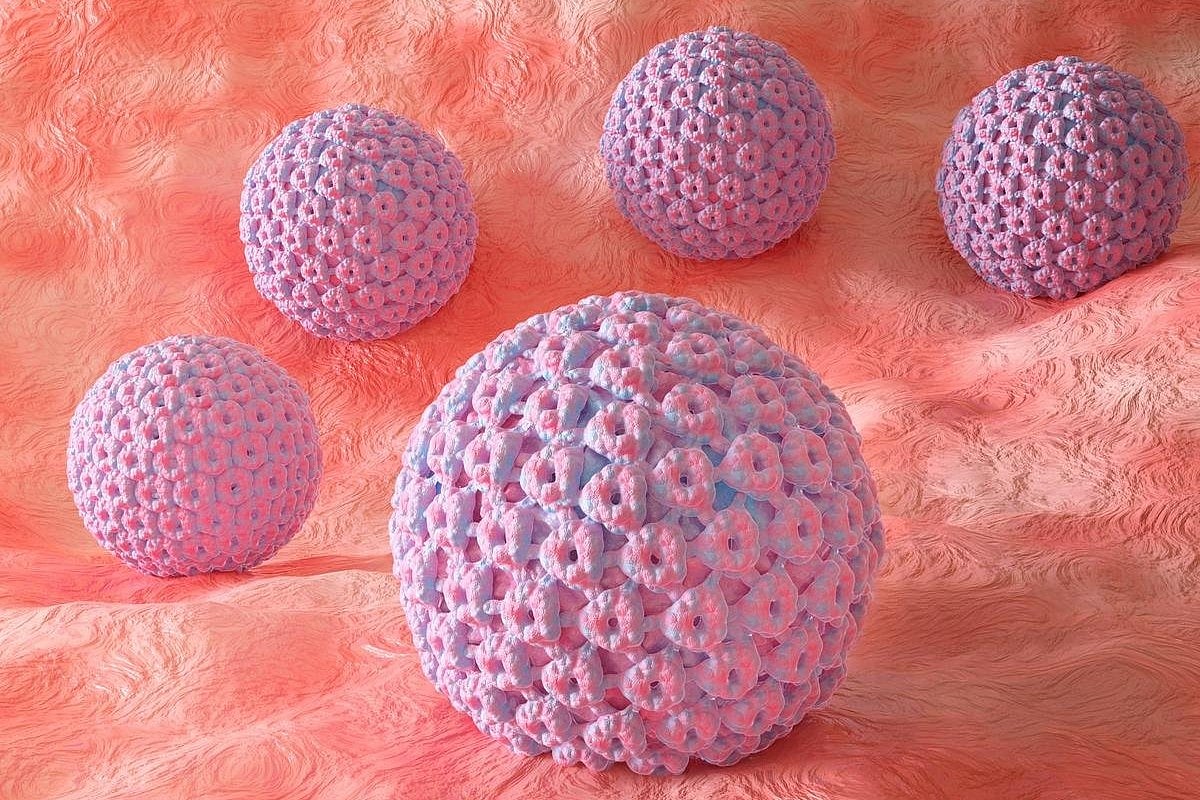
First-Ever Detection For Early Head And Neck Cancers Under Development
A new blood test can potentially detect head and neck cancers caused by human papilloma virus (HPV) up to 10 years before symptoms appear, a new study says.
The test, called HPV-DeepSeek, detects microscopic fragments of HPV DNA that have broken off from a tumor and entered the bloodstream, researchers reported Sept. 10 in the Journal ...
- Dennis Thompson HealthDay Reporter
- |
- September 12, 2025
- |
- Full Page
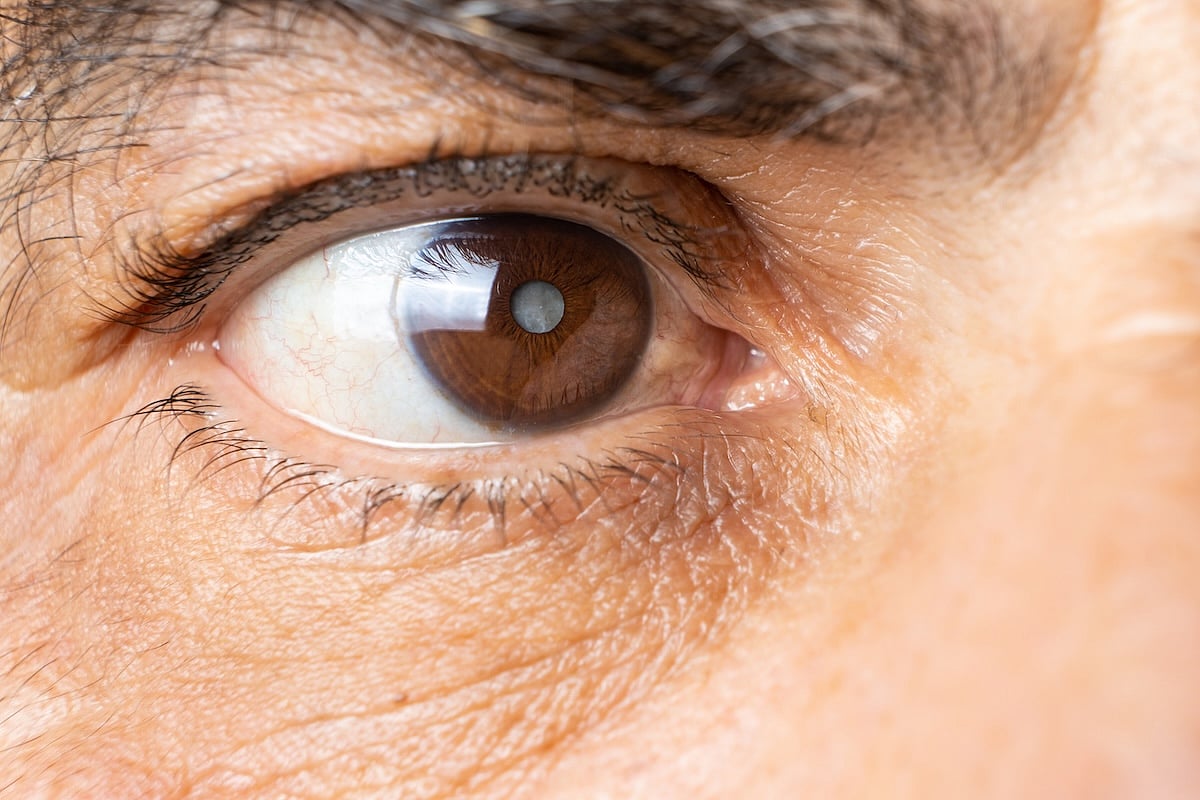
Same-Day Cataract Surgery On Both Eyes Safe And Effective
Same-day cataract surgery on both eyes can be safe, effective and practical, according to a pair of new studies.
Cataract surgery typically is performed on one eye then the other, with procedures scheduled weeks or months apart.
But getting surgery done on both eyes at once works just as well, and doesn’t appear to interfere wi...
- Dennis Thompson HealthDay Reporter
- |
- September 12, 2025
- |
- Full Page
Even Healthy Babies At Risk For Severe RSV Infection
RSV can make even healthy, full-term babies sick enough to land in the hospital or require intensive care, a new study says.
In fact, more than half of RSV-infected infants and children who required intensive care or prolonged hospitalization were born at term and had no chronic health problems, researchers reported Sept. 9 in The Lanc...
- Dennis Thompson HealthDay Reporter
- |
- September 12, 2025
- |
- Full Page
High THC Levels May Affect Egg Quality and Fertility, New Study Finds
High levels of THC — the compound in marijuana that causes a “high” — may affect how eggs develop and could lead to fertility problems, miscarriages and chromosome issues in embryos, new research shows.
The findings were reported Sept. 9 in the journal Nature Communications.
Researchers analyzed more ...
- I. Edwards HealthDay Reporter
- |
- September 11, 2025
- |
- Full Page
COVID-19 Falls From Top 10 U.S. Causes of Death for First Time Since 2020
For the first time since the pandemic began, COVID-19 dropped out of the nation’s top 10 causes of death in 2024, new U.S. government data shows.
Overall U.S. death rates also fell to their lowest level since 2020, according to the National Center for Health Statistics.
Heart disease and cancer remained the leading causes...
- I. Edwards HealthDay Reporter
- |
- September 11, 2025
- |
- Full Page
Endangered Shark Meat Found in U.S. Grocery Stores, Study Says
Shark meat sold in the U.S. may come from endangered species without consumers realizing it, a new study warns.
Researchers tested 29 shark products, including 19 fillets bought in grocery stores and seafood markets across North Carolina, Florida, Georgia and Washington, D.C., plus 10 jerky products purchased online.
Using DNA ...
- I. Edwards HealthDay Reporter
- |
- September 11, 2025
- |
- Full Page
What to Know About Anaphylaxis: Symptoms, Triggers & Treatment
When people hear about anaphylaxis, they often associate the severe allergic reaction with food allergies. And while food allergies can cause anaphylaxis, allergies to insect stings, medications and latex can also trigger the response.
Anaphylaxis is rare. In most cases, people with allergies develop mild to moderate symptoms, such as wate...
- Todd A. Mahr, MD, Executive Medical Director, American College Of Allergy, Asthma And Immunology HealthDay Reporter
- |
- September 11, 2025
- |
- Full Page
Parent-Focused Prevention Programs For Childhood Obesity A Bust, Evidence Says
Programs that recruit parents to help fight early childhood obesity aren’t doing much good at all, a new evidence review says.
There’s no evidence that these programs have any impact on the body mass index (BMI) of toddlers, researchers reported in The Lancet. BMI is an estimate of body fat based on height and weight.<...
- Dennis Thompson HealthDay Reporter
- |
- September 11, 2025
- |
- Full Page
Air Pollution Might Accelerate Alzheimer's Progression
Air pollution might speed up the devastating effects of Alzheimer’s disease on the brain, a new study says.
Alzheimer’s patients exposed to higher levels of particle pollution appeared to suffer an increased buildup of toxic amyloid and tau proteins in their brains, researchers reported Sept. 8 in JAMA Neurology.
- Dennis Thompson HealthDay Reporter
- |
- September 11, 2025
- |
- Full Page
Food Deserts Potentially Deadly For People With Irregular Heartbeat
Living in a food desert can be potentially deadly to people with irregular heart beats, a new study says.
Patients with atrial fibrillation have a doubled risk for stroke and nearly quadrupled risk of early death if they live in a neighborhood without a nearby full-service grocery store, researchers reported in the journal JACC: Advanc...
- Dennis Thompson HealthDay Reporter
- |
- September 11, 2025
- |
- Full Page
How To Help Your Back-To-School Kid With Their Allergies
Back-to-school is an exciting time for kids and parents, but it also marks the beginning of fall allergies as well as the cold and flu season.
So on top of gathering clothes and books and school supplies, parents also might have to figure out why their child has a runny nose.
“It is very confusing because you're like, what's ha...
- Dennis Thompson HealthDay Reporter
- |
- September 11, 2025
- |
- Full Page
Insomnia Might Increase Dementia Risk Among Seniors
Your beauty rest might be protecting your brain health as well, a new study says.
Seniors with chronic insomnia might experience a faster decline in their memory and thinking skills as they age, researchers reported today in the journal Neurology.
Chronic insomniacs had a 40% higher risk of developing dementia or suffering a...
- Dennis Thompson HealthDay Reporter
- |
- September 11, 2025
- |
- Full Page
Household Items Tied to Sharp Increase in Self-Harm Cases in Teens
Cases of self-harm involving 6- to 12-year-olds have risen sharply, and researchers warn that common household products are often involved.
Their findings appear in the Sept. 8 issue of Pediatrics, the journal of the American Academy of Pediatrics.
Researchers analyzed more than 1.5 million reports made to U.S. poison contro...
- I. Edwards HealthDay Reporter
- |
- September 10, 2025
- |
- Full Page
Climate Change Could Be Fueling Higher Sugar Consumption
Even as the U.S. heads into cooler months, climate change is driving hotter overall temperatures, and new research suggests that rising heat is pushing Americans to consume more sugary drinks and frozen treats — with some potential health risks.
Researchers analyzed food purchases from U.S. households between 2004 and 2019 and compar...
- I. Edwards HealthDay Reporter
- |
- September 10, 2025
- |
- Full Page





.jpg?w=1920&h=1080&mode=crop&crop=focalpoint)