Get Healthy!
Staying informed is also a great way to stay healthy. Keep up-to-date with all the latest health news here.
24 Mar
Unsafe Levels of Lead Found in Kids' Fast Fashion
Researchers tested 11 shirts from children's fast-fashion and discount clothing stores. All contained lead levels above FDA limits, according to the results.
23 Mar
Stopping GLP-1 Drugs May Raise Heart Risks
A new study finds even brief breaks from GLP-1 medications can increase the risk of heart attack, stroke, and death—highlighting the importance of staying on treatment.
20 Mar
High Blood Pressure Deaths On the Rise in Younger Women
A new study finds deaths from heart disease related to high blood pressure have quadrupled over the past two decades in young women.
Baby Walkers Sold on Amazon Recalled Over Fall Risk
About 90 infant walkers sold online are being recalled because they may not protect babies from falling.
The recall involves Goregent Infant Walkers, which do not meet federal safety rules, the U.S. Consumer Product Safety Commission (CPSC) said.
Officials say the walkers can fit through standard doorways and may fail to stop a...
- HealthDay Staff HealthDay Reporter
- |
- March 25, 2026
- |
- Full Page
HelloFresh Pizza Recall Issued in 10 States Over Metal Risk
A frozen pizza sold through the popular meal delivery service HelloFresh is being recalled after reports that it may contain small metal fragments.
The U.S. Food and Drug Administration (FDA) said the recall is for the Basil Pesto & Mozzarella pizzas made by Bakkavor, a company based in Charlotte, North Carolina.
The FDA has labe...
- HealthDay Staff HealthDay Reporter
- |
- March 25, 2026
- |
- Full Page
Want To Protect Your Brain? Science Says Exercise
Lunges and squats may do more for the brain than previously thought, including helping protect it from damage tied to aging and dementia, a new study suggests.
In research published this month in the journal Cell, scientists found that physical activity may help repair the blood-brain barrier, a layer of cells that protects the br...
- HealthDay Staff HealthDay Reporter
- |
- March 25, 2026
- |
- Full Page
Inclusive High Schools Benefit All Students, Not Just LGBTQ Teens
An inclusive, welcoming environment for LGBTQ teenagers helps all students attending high school, a new study says.
Cisgender students (those whose gender identity matches the sex they were assigned at birth) experience an increase in their own social well-being if they attend a school that promotes openness and inclusivity toward LGBTQ te...
- Dennis Thompson HealthDay Reporter
- |
- March 25, 2026
- |
- Full Page
Childhood Obesity Undercuts The American Dream For Some, Study Says
Childhood obesity could be robbing some kids of their chance at the American Dream.
Children who are obese are far less likely to climb the economic ladder as adults, compared to kids who have a normal weight, researchers recently reported in the Journal of Population Economics.
In essence, kids’ excess weight appears ...
- Dennis Thompson HealthDay Reporter
- |
- March 25, 2026
- |
- Full Page
U.S. Medicine, Science Facing An Online Misinformation Siege, Poll Concludes
Americans are trapped in a ruthless social media spin cycle of viral misinformation regarding science and medicine, with many actively contributing to the problem, a new Harris Poll has found.
People have come to depend on social media for news about health and science, but the poll found they also blame these platforms for the spread of m...
- Dennis Thompson HealthDay Reporter
- |
- March 25, 2026
- |
- Full Page
'Universal Donor' Blood Supplies Dangerously Low, Study Warns
Blood banks are reporting dangerously low supplies of “universal donor” O-negative blood, a new study warns.
O-negative blood can be given to patients of any blood type, making it essential in medical emergencies where a person’s blood type is unknown, researchers said.
But O-negative supplies have dropped to critic...
- Dennis Thompson HealthDay Reporter
- |
- March 25, 2026
- |
- Full Page
Parental Loss Due to Drugs, Violence Raises Child Death Risk by 2,000%
When a parent dies unexpectedly due to violence, self-harm or addiction, the trauma for a child left behind is immense.
And a new study reveals that this heartbreak carries more than just emotional weight — it significantly increases the child's own risk of dying before they reach adulthood.
Researchers at the Unive...
- Deanna Neff HealthDay Reporter
- |
- March 25, 2026
- |
- Full Page
Why Stepping Outside May Help You Eat Better
Most people visit a local park to clear their heads or get some exercise, but a new study suggests that green space might be a secret weapon for a better diet.
Drexel University resesearchers found there is a powerful link between the time we spend in nature and the quality of the food we put on our plates.
The study — publishe...
- Deanna Neff HealthDay Reporter
- |
- March 25, 2026
- |
- Full Page
TrumpRx Adds Diabetes, COPD Drugs at Steep Discounts
TrumpRx, the president's discount program for prescription drugs, is adding three more products to its site as it continues efforts to lower drug prices.
The newly added medicines include two for type 2 diabetes, Jentadueto and Jentadueto XR, and one for chronic obstructive pulmonary disease (COPD), Striverdi Respimat.
All thre...
- HealthDay Staff HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
New Lyme Disease Vaccine Shows Strong Results in Trial
A new vaccine meant to prevent Lyme disease may be one step closer to approval.
Drugmaker Pfizer and French vaccine company Valneva said their vaccine, called LB6V, worked more than 70% of the time in a clinical trial to prevent Lyme disease in people ages 5 and older.
The companies also said the vaccine was well tolerated, with no s...
- HealthDay Staff HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
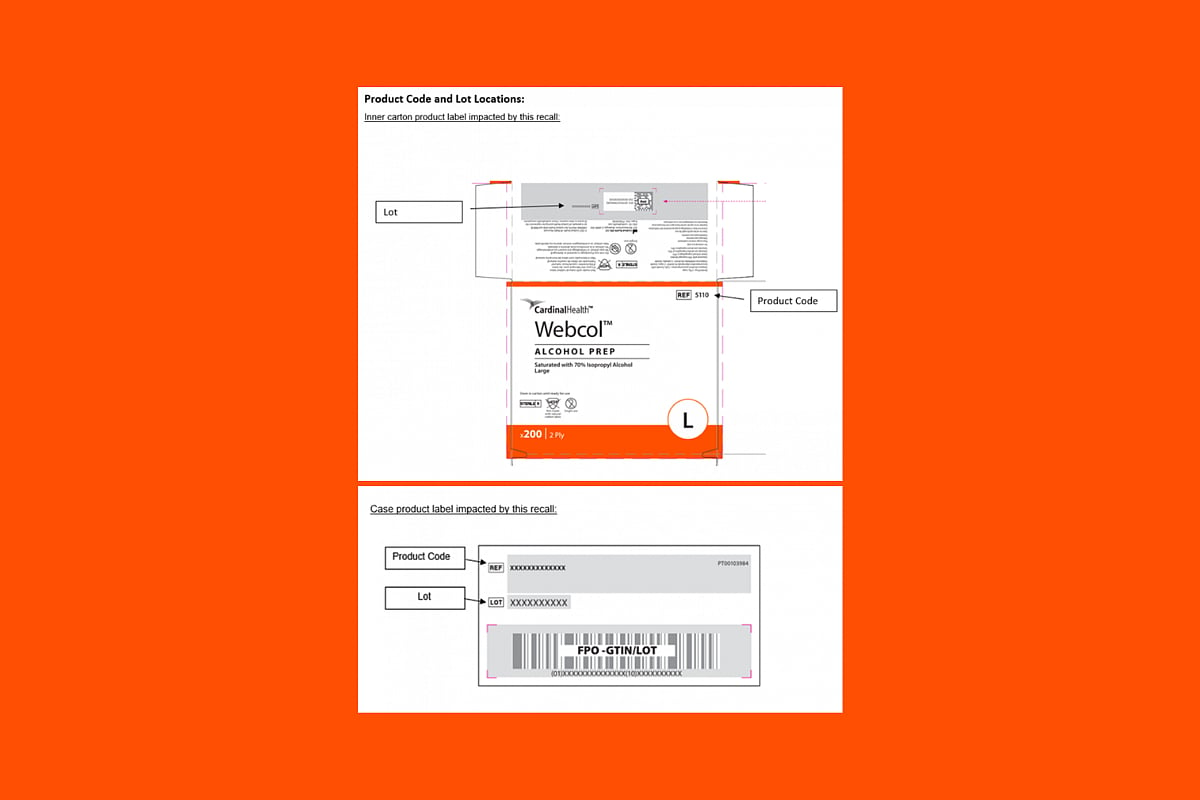
Alcohol Prep Pads Recalled Over Bacteria Risk, Cardinal Health Says
Cardinal Health is recalling some of its alcohol prep pads after tests found possible bacterial contamination that could lead to infections.
The company said certain lots of its Webcol Large Alcohol Prep Pads (70% isopropyl alcohol) may contain the bacteria Paenibacillus phoenicis.
These pads are commonly used to clean the s...
- HealthDay Staff HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
Study Warns Fluoride Bans May Raise Tooth Decay in Children
Removing fluoride from drinking water could lead to more cavities in kids and higher health care costs, a new analysis suggests.
Researchers estimate that if five states stop adding fluoride to public water, more than 132,000 additional kids could need dental treatment within three years.
That includes procedures like fillings ...
- HealthDay Staff HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
Your Choice Of Booze Influences Your Risk Of Death, Study Says
Too much alcohol of any type is bad for a person’s health, but some booze is more harmful than others, a new study says.
Beer, cider and liquor all appear to increase people’s risk of an early death, even at low levels of imbibing, researchers are slated to report Saturday at a meeting in New Orleans of the American Colle...
- Dennis Thompson HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
Insulin Prices Fell For Medicare Patients Under Biden-Era Caps, Study Finds
Biden-era policies have reined in insulin prices for Medicare patients, a new study says.
Roughly 3 out of 4 (75%) Medicare prescription beneficiaries were paying $35 or less for each month’s supply of insulin within a year of price caps enacted under President Joe Biden, researchers reported in the Journal of the American Medica...
- Dennis Thompson HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
AI Gets a 'D' When Judging Scientific, Medical Claims
Folks who rely on chatbots for their scientific and medical info, be forewarned — artificial intelligence (AI) gets a "D" when it’s asked to evaluate whether a claim is true or false, a new study says.
ChatGPT’s accuracy in assessing scientific claims was only about 60% better than random guessing, a score that would earn...
- Dennis Thompson HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
New Fathers Face Mental Health Challenges, Study Finds
New fathers might be proud poppas, but their mental health might be shaky as they adapt to their increased responsibilities, a new study says.
Men have a 30% increased risk of depression and stress disorders by the end of their first year of fatherhood, researchers reported March 23 in JAMA Network Open.
This follows a menta...
- Dennis Thompson HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
New Online Tool Helps Parkinson's Patients Weigh Brain Implant Decision
Brain implants are proven to help Parkinson’s disease patients control their symptoms, but deciding whether to go through with such an invasive surgery can be overwhelming.
Now, researchers have developed an online decision-making tool to help patients understand the implants and boost their confidence, according to recent report in ...
- Dennis Thompson HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
Cheap Children's Clothing Tainted With Lead, Study Says
“Fast fashion” is an affordable way for parents to keep up with their kids’ growth spurts, but these cheaper duds might come with real health risks, a new study says.
The fabric in some fast-fashion shirts contains high levels of lead, researchers reported Monday at the spring meeting of the American Chemical Society in A...
- Dennis Thompson HealthDay Reporter
- |
- March 24, 2026
- |
- Full Page
Teens Often Pressured To Send Sexual Photos by Someone They Know, Study Finds
Many teens who are asked to send sexual photos are being pressured by someone they know, and most often, it's a boyfriend or girlfriend, according to new research.
The study, published March 17 in JAMA Network Open, asked more than 6,200 young people ages 18 to 28 to look back on experiences they had before age 18.
Researche...
- HealthDay Staff HealthDay Reporter
- |
- March 23, 2026
- |
- Full Page